
Note that chlorine atom (2.8.7) gains electron to its valance shell to achieve a stableĮlectron arrangement.

½ Cl2 (g) → Cl (g)įor the formation of calcium chloride, two Cl (g) is required, therefore total atomisation enthalpy will be double that is 242 kJ/mol.Įlectron affinity of chlorine: This is the amount of energy released during the addition of electron in an isolated neutral gaseous chlorine atom. The reaction enthalpy is half of the bond dissociation enthalpy of chlorine. Ca (g) → Ca+(g) + e+Ītomization enthalpy of Chlorine: This step involves dissociation of Cl2 (g) into Cl(g) atoms. Note that calcium atom (2.8.8.2) loses electron to achieve a stable electron arrangement and stable state has less energy when compared to other state, thus energy is released as calcium atom loses electrons to form gaseous -2 ion. Hence the second ionization energy for calcium ion is 1145 kJ/mol. The removal of second electron requires more energy as it is difficult to remove electron from a cation. Ionisation enthalpy of calcium: Calcium forms di-positive ion (Ca2+), therefore the energy involve in the removal of first electron is called as first ionization energy and valued 590 kJ/mol. Note that solid calcium absorbs heat energy to form gaseous calcium, thus the products have higher energy than the reactants, this is an exothermic reaction. Ītomization enthalpy of calcium: This step involves the conversion of solid calcium to gaseous state.
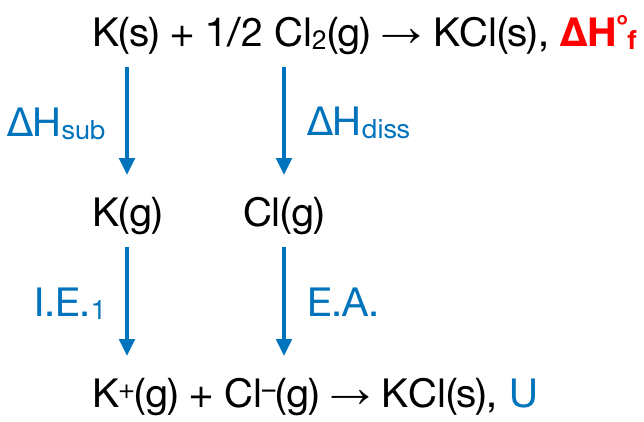
Note: Always remember that in the Born-Haber cycle, the word cycle refers to the fact that one can also equate to zero the total enthalpy changes for a cyclic process.Born Haber Cycle, Example:CaCl2 The Born Haber cycle for the formation of calcium chloride form its constituent elements involves following steps. The amount of energy released when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion is known as electron affinity.įrom the above discussion, it is clear that both (A) and (B) are correct. The energy required to remove one or more electrons to make a cation is a sum of successive ionization energies. If the element is normally a molecule then we first have to consider its bond dissociation enthalpy.

To make gaseous ions from elements it is necessary to turn each element into gaseous atoms and then to ionize the atoms. A Born–Haber cycle applies Hess's law (The change of enthalpy in a chemical reaction is independent of the pathway between the initial and final states.) to calculate the lattice enthalpy by comparing the standard enthalpy change of formation of the ionic compound (from the elements) to the enthalpy required to make gaseous ions from the elements. The lattice energy is the enthalpy change involved in the formation of an ionic compound from gaseous ions, or sometimes defined as the energy to break the ionic compound into gaseous ions. The first question that comes to our mind is what is the Born-Haber cycle?īorn-Haber cycle is a cycle of enthalpy change of process that leads to the formation of a solid crystalline ionic compound from the elemental atoms in their standard state and of the enthalpy of formation of the solid compound such that the net enthalpy of the reaction becomes zero.īorn–Haber cycles are used primarily as a means of calculating lattice energy, which cannot otherwise be measured directly. It was named after two German scientists Max Born and Fritz Haber, who developed it in 1919. Hint: The Born–Haber cycle is an approach to analyze the energy of a given reaction.
